Earlier in the year, I had LDR (low-dose) brachytherapy treatment for prostate cancer. The way it works is that the radiation oncologist, in concert with the urologist/surgeon, maps out where the cancer is in the prostate, and then builds up a map in 3D of exactly where to implant a set of radioactive seeds to irradiate the cancer and as little as possible of other things, like the bladder, urethra, and colon.
The treatment can use various radioactive isotopes; in my case, we decided on Palladium-103, which has a half-life of a tiny bit less than 17 days, and decays by electron capture, which I had not previously heard of.
One of the K-shell electrons in palladium-103 has has a chance of having nonzero probability density inside the nucleus. (Think of a big cartoon sign pointing to the nucleus that says “YOU MIGHT BE HERE” for the electron.)
If that happens, there’s a possibility that the weak nuclear force interaction between the electron and a proton in the nucleus will convert that proton into a neutron. That transforms the atom from palladium to rhodium and emits a neutrino.
No big deal to emit a neutrino; billions of them are constantly sleeting through us every second from the sun. But! Now the rhodium atom is missing an electron in the K-shell, so one of the existing electrons drops into that shell and now the atom has excess energy to dump. One of two things happens:
- The “we’ve all seen this one in physics class”: the atom emits a photon (in this case a low-energy X-ray), and we’re back to normal energy. Ho hum.
- Then there’s the “you can do that?” option: the electrons just play “hot potato” and pass around the extra energy until one is bound loosely enough to be kicked out — this is an Auger electron (named after Pierre Victor Auger, though Lise Meitner published it a year earlier — the guys get the credit again); from the radiomedical standpoint, it acts as if it were a beta particle — it’s a high-energy electron — but doesn’t come from a nuclear decay: the electron is literally handed the excess energy and sent packing with it.
For treating cancer, both of these are good news: the Auger electron is very short range but has high interactivity with the cancer cells to put them on the Oblivion Express (okay, that’s Brian Auger, not Pierre!); the X-ray photons travel further, but aren’t as strong. This means that the radioactivity is concentrated right where it’s needed.
But it’s not 100% absorbed.
One of the warnings I got was to make sure that I stayed around six feet away from young children and possibly-pregnant women for the first six weeks, as those are folks who can be affected much more by even the weak radioactivity I was shedding.
That made me wonder: just how radioactive was I, compared to when I started? Let’s make a chart!
Fortunately palladium-103’s decay is super simple: one path to rhodium-103, which is stable, so I can use the basic decay-curve equation to figure out exactly how much Pd-103 is left over time[1].
This only requires us to know the decay constant λ, which we do: 16.99 days. We can plug that into a little Python program and get a nice curve:

So the breakdown is actually pretty fast! We’re nearly at zero after 20 weeks, but because it’s an exponential curve, it’s a bit hard to read off numbers. Let’s look at that as a table:
Weeks | % Remaining
----------------------
0.0 | 100.0000
2.0 | 56.4887
4.0 | 31.9097
6.0 | 18.0254
8.0 | 10.1823
10.0 | 5.7519
12.0 | 3.2492
14.0 | 1.8354
16.0 | 1.0368
18.0 | 0.5857
20.0 | 0.3308
22.0 | 0.1869
24.0 | 0.1056
26.0 | 0.0596
28.0 | 0.0337
30.0 | 0.0190
32.0 | 0.0107
34.0 | 0.0061
36.0 | 0.0034
38.0 | 0.0019
40.0 | 0.0011
42.0 | 0.0006
44.0 | 0.0003
46.0 | 0.0002
48.0 | 0.0001
50.0 | 0.0001
52.0 | 0.0000
So at 6 weeks, the “it’s okay to stop warning people” cutoff, I’m at about 18% of the original intensity. That doesn’t give me an absolute number, but is interesting.
I posted this on Reddit in r/ProstateCancer, just because it interested me; the mods did remove it, and fair, it’s more a curiosity than anything useful. Before it got pulled, though, I had one person ask me about how fast Pluvicto decayed — that’s Lutetium-177, and again, very fortunately a one-step-to-stable path. λ for Lu-177 is much shorter, about 6.4 days, so the curve falls off much faster:
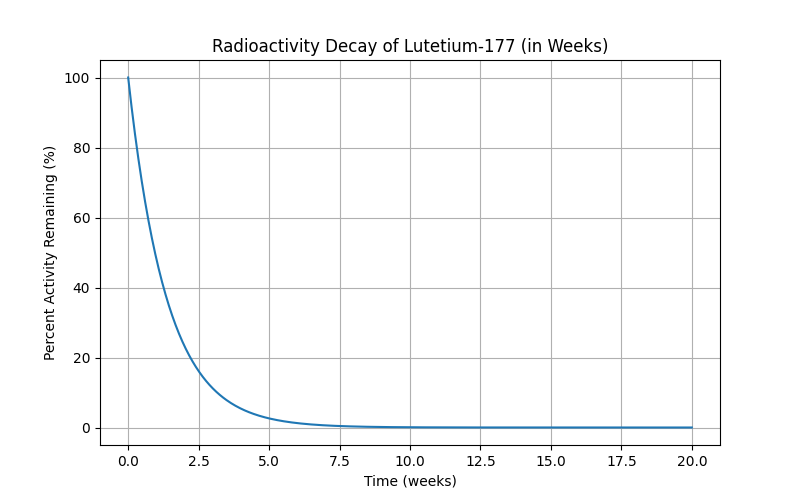
We’re pretty much at zero after ten weeks, which is significantly faster; the table looks like this:
Weeks | % Remaining
----------------------
0.0 | 100.0000
2.0 | 23.2255
4.0 | 5.3943
6.0 | 1.2528
8.0 | 0.2910
10.0 | 0.0676
12.0 | 0.0157
14.0 | 0.0036
16.0 | 0.0008
18.0 | 0.0002
20.0 | 0.0000
The warnings are different for Lu-177: “limit close contact (less than 3 feet) with household contacts for 2 days or with children and pregnant women for 7 days. Refrain from sexual activity for 7 days, and sleep in a separate bedroom from household contacts for 3 days, from children for 7 days, or from pregnant women for 15 days.”
The remaining active Lu-177 is at about the same level, 20-ish percent in that amount of time, so my intuitive guess is that in terms of “radiation exposure to others”, the two are about the same.
Pd-103 hangs around longer, but because it’s just dropping those Auger electrons and the low-energy X-rays, they don’t propagate as much, and the effect is much more localized.
Lu-177 in Pluvicto circulates through the entire body, and binds to metastatic cancer cells there, so it makes sense that we’d want something that decayed a lot faster. (The Lu-177 decay is gamma and actual beta emission.)
Conclusions? None really, it was simply trying to understand what was happening better, and was probably displacement activity. 🙂
If you want to see the Python program that made the charts and tables, check out https://github.com/joemcmahon/decay_curve.
[1] In college, I wrote my very first computer program to simulate the decay of a single U-235 atom to a stable state. I had learned about if statements and the rand() function, but not arrays. So I had a sheaf of if‘s that figuratively “ticked the clock” by one half life for each if block, moving on to the next when the random coin flip said the atom had taken another step along the decay path. It had all the structure and sophistication of a noodle.
This was essentially a very bad and ridiculously unsophisticated Monte Carlo simulation, but in my defense, I had never written a computer program at all before, and I was extra proud I managed to make it work.
There were a lot of long printouts of decay timelines on fanfold paper.
Leave a Reply
You must be logged in to post a comment.